Surface Structure of Scherzo C18 Stationary Phase and Mobile Phase Design
- A World-First Innovative Concept / ODS + Anion + Cation Ligands / Control of Ligand Ion Amount -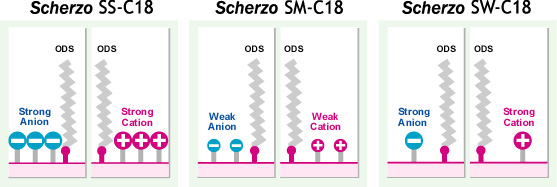
Scherzo C18 family includes three types of columns. All three columns share a common structure of "ODS + anion ligand + cation ligand." However, each stationary phase differs in the strength and amount of ion ligands introduced, resulting in significant differences between columns in terms of the retention of ionic compounds.
Scherzo SS-C18
An ODS stationary phase with a high loading of strong ion ligands is highly effective for retaining ionic compounds that could not be retained by conventional ODS.
It is particularly useful for retaining solutes with relatively weak charges, such as zwitterions and weak cations.
- Example of Amadori Compounds
Mobile Phase A: water / formic acid = 100 / 0.3
Mobile Phase B: acetonitrile / 100mM ammonium formate = 20 / 80Retention is achieved through both ion-exchange, by changing pH and ionic strength from formic acid to ammonium formate, and hydrophobic interactions by increasing organic solvent concentration.
- Example of Shikimic Acid and Malic Acid
Mobile Phase A: 10mM ammonium formate
Mobile Phase B: water / acetonitrile / formic acid = 75 / 25 / 0.5For weak ionic carboxylic acids, retention is achieved by ionic adsorption under neutral and low ionic strength conditions, and desorption is facilitated by ion-exchange through lowering the mobile phase pH (exchanging with formate ions from formic acid).
>>>See all Scherzo SS-C18 Application Database <<<
Scherzo SW-C18
An innovative ODS stationary phase with a small amount of strong ion ligands is effective for solutes with relatively strong charges that are difficult to elute with SS-C18.
- Example of Strong Organic Base Catalysts
Mobile Phase A: water / formic acid = 100 / 0.1
Mobile Phase B: acetonitrile / 100mM ammonium formate = 60 / 40Retention is achieved through both ion-exchange, by changing pH and ionic strength from formic acid to ammonium formate, and hydrophobic interactions by increasing organic solvent concentration. This is effective when retention with SS-C18 is too strong, making elution difficult.
- Example of Orexin (Peptide) in LC-MS Analysis
Mobile Phase A: water / formic acid = 100 / 0.1
Mobile Phase B: 150mM ammonium formate / acetonitrile = 40 / 60Adsorption occurs in acidic conditions (formic acid) below the isoelectric point, and desorption is facilitated by increasing pH, ionic strength, and organic solvent concentration for both ionic and hydrophobic interactions.
>>>See all Scherzo SW-C18 Application Database <<<
Scherzo SM-C18
Scherzo SM-C18 is an ODS stationary phase with moderate introduction of weak ion ligands. It is designed to moderately retain both acidic and basic substances in neutral mobile phases, making it versatile. It is also effective for strong cations such as quaternary ammonium compounds. It is designed for comparison with Unison UK-C18, allowing for understanding of retention and separation characteristics with and without ionic interactions.
- Example of Amlodipine Besylate (Antihypertensive Drug)
Mobile Phase A: 10mM ammonium formate
Mobile Phase B: 100mM ammonium formate / acetonitrile = 30 / 70Both cations (amlodipine) and anions (besylate) can be analyzed simultaneously through ion-exchange (adsorption and desorption) by varying the ionic strength of ammonium formate. Compared to other Scherzo C18 columns, its weaker ionic interactions result in moderate retention and relatively sharp peaks.
- Example of Benzalkonium Chloride
Mobile Phase A: 10mM ammonium formate
Mobile Phase B: methanol / formic acid = 100 / 0.5For strong cationic compounds like quaternary ammonium, adsorption occurs under neutral pH and low ionic strength conditions, and desorption is achieved ion-exchange by lowering the pH to suppress dissociation of weak anion ligands.
- Example of Mevalonic Acid and Lactone
Mobile Phase: 5mM ammonium formate / acetonitrile = 90 / 10
Using Scherzo SM-C18, mevalonic acid interacts with the cationic stationary phase and is retained under neutral pH and low ionic strength conditions. In contrast, mevalonic acid elutes quickly with Unison UK-C18, which lacks ion-exchange interaction. Mevalonolactone, being non-ionic, shows similar hydrophobicity to the UK-C18. By analyzing under the same neutral pH conditions, it is possible to determine whether the solute is ionic or not.
>>>See all Scherzo SM-C18 Application Database <<<
Elution Behavior of Non-Ionic Compounds
For non-ionic substances without ion-exchange interactions, the three types of Scherzo C18 exhibit similar elution behavior.
For Organic Acids, Intrada Organic Acid Column is Recomennded
For compounds difficult to analyze with organic acids, inorganic acids, or organophosphates, the LC-MS specific Intrada Organic Acid column is recommended.
For Amino Acids, Intrada Amino Acid Column is Recommended
For amino acids and their related compounds, the LC-MS specific Intrada Amino Acid column is recommended.